[Please refer to the instruction manual.]
[General Name] Automatic Electronic Blood Pressure Monitor
[Product Name] Omron Wrist Blood Pressure Monitor HEM-6160 Series
[Contraindications/Prohibitions]
(1) Self-diagnosis of measurement results or treatment based on self-diagnosis [May lead to worsening of symptoms.]
(2) Measurement on an injured or injured arm [May lead to worsening of symptoms.]
(3) Measurement on an arm receiving intravenous infusion or blood transfusion [May lead to worsening of symptoms.]
(4) Use near flammable or combustible gases [May cause ignition, fire, or explosion.]
(5) Use beyond the service life [May not measure correctly.]
(6) Use by an unspecified number of people in medical institutions or public places [May cause accidents or problems.] ]
[Specifications]
External dimensions: Approx. 84 (width) x 62 (height) x 21 (depth) mm (excluding cuff)
Weight: Approx. 85 g (excluding batteries)
Operating environment conditions: +10 to +40℃ / 15 to 90% RH (non-condensing) / 800 to 1060 hPa
Wrist circumference: 13.5 to 21.5 cm
[Electrical ratings]
Power supply: 2 x AAA alkaline batteries (DC 3V 3.0W)
Electric shock protection: Internally powered equipment
BF type applied part: Cuff
IP protection rating: IP22
[Standards related to performance and safety]
(1) Error in blood pressure measurement by clinical performance test:
• Average error with auscultation method: within ±5 mmHg
• Standard deviation: within 8 mmHg
(2) Pressure display error due to environmental conditions:
• Pressure range 0 to 299 mmHg; within ±3 mmHg
(3) Stopping operation:
• Time required to rapidly deflate from 260 mmHg to 15 mmHg; 10 seconds or less
(4) Blood pressure measurement reproducibility: 3 mmHg or less
(5) Maximum cuff pressure: 299 mmHg
[Purpose of use or effect]
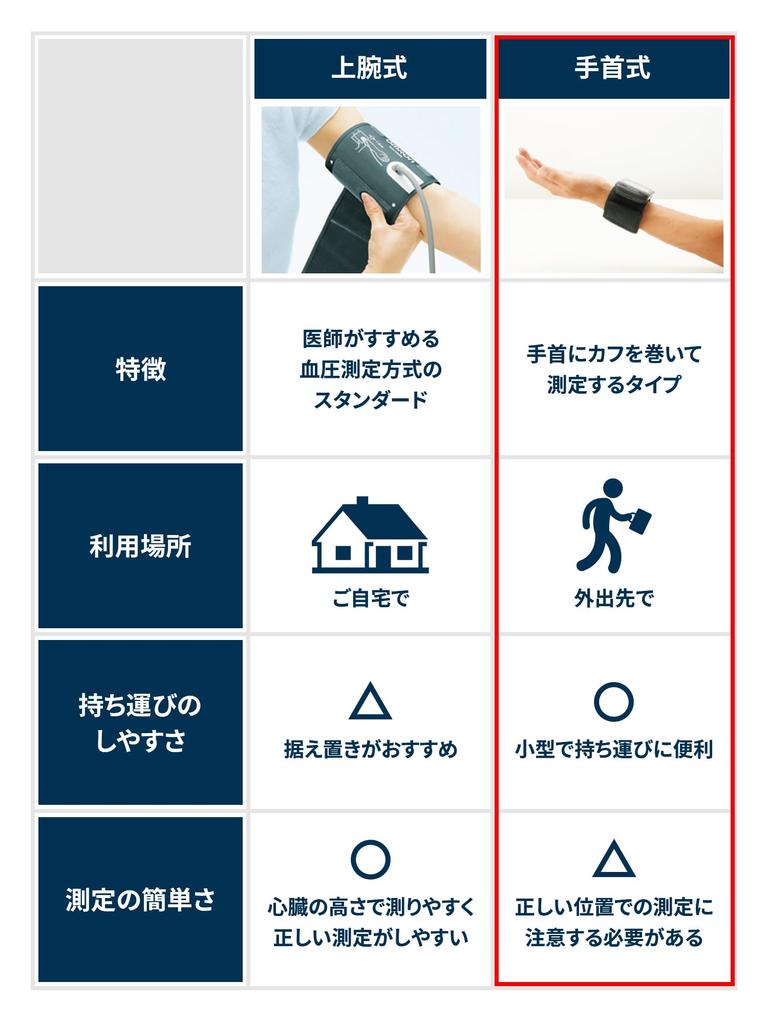
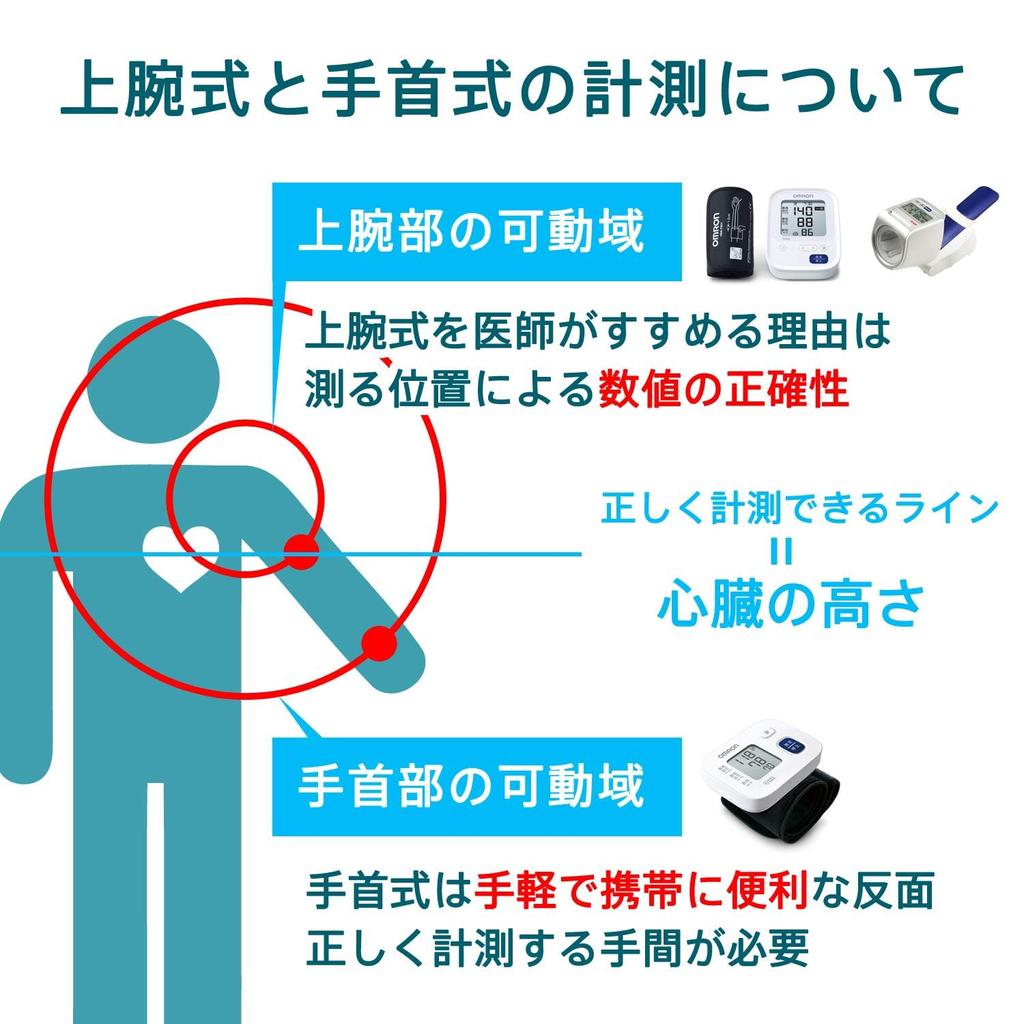
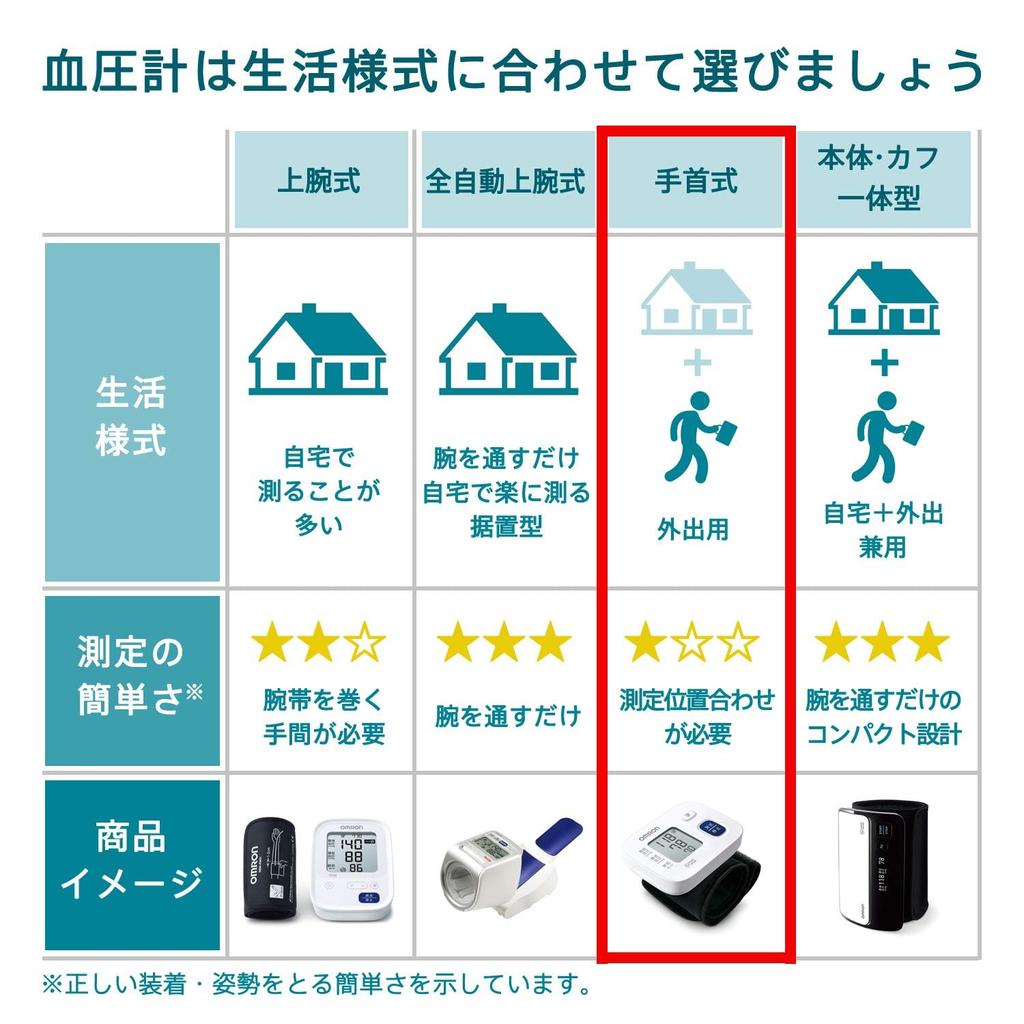
To measure systolic and diastolic blood pressure non-invasively for home health management.
[Precautions for use]
1. Individuals with severe circulatory disorders or blood diseases should use under the guidance of a physician.
2. Temporary internal bleeding may occur due to compression.
3. Do not use on infants or individuals unable to express their wishes.
4. Do not use for purposes other than blood pressure measurement.
5. Do not disassemble or modify the main unit or dedicated cuff.
6. Insert the battery correctly, ensuring the positive (+) and negative (-) terminals are correctly aligned.
7. Use the specified battery.
8. Remove the battery when not in use for an extended period.
[Manufacturer and Distributor Information]
Manufacturer and Distributor: Omron Healthcare Co., Ltd.
Phone: 0120-30-6606
(Omron Customer Service Center)
Specified Controlled Medical Device Medical Device Certification (Approval) Number: 230AGBZX00001000
Wrist Type
Applicable Wrist Circumference: 13.5~21.5cm
External Dimensions: Approx. 84mm (W) x Approx. 21mm (D) x Approx. 62mm (D)
Power Supply: Dry Cell Battery